
肥胖与新型冠状病毒病肺炎(Covid-19)导致的重症、死亡风险增加独立相关。体重增加引起的病理生理变化导致代谢功能障碍、慢性炎症、免疫反应受损和多系统疾病,增加了Covid-19严重性。肥胖和Covid-19潜在机制是什么?
发表在Trends in Endocrinology & Metabolism 的一篇综述[1]对此进行了阐述,并且提出时下较为流行的进食方案,间歇性禁食(intermittent fasting)也称为轻断食,这种进食方案能否改善代谢健康和免疫功能,降低Covid-19加重的风险?这篇综述也进行了很好地回答。
Covid-19感染与肥胖:两大流行的交叉
Covid-19俗称“新冠肺炎”,来自多个临床队列的研究表明,肥胖与SARS-CoV-2感染的并发症、疾病的严重程度和死亡率的增加相关[2-4]。一项对美国90多万新冠肺炎住院患者的风险评估分析发现,近三分之二的住院患者合并心脏代谢性疾病,即肥胖、糖尿病(DM)、高血压和心力衰竭,肥胖占住院总数的30%[5]。据报道, 与非肥胖患者(BMI<25 kg/m2)相比,体重指数(BMI)升高的患者对侵入性机械通气的需求显著增加,更有可能发展为严重肺炎,表现出更严重的肺部病理变化和损伤,死亡率风险增加,而这些独立于年龄、性别、糖尿病和高血压 [4]。
与新冠肺炎的快速传播不同,几十年来肥胖呈现缓慢的大流行趋势。1975年至2016年,全球肥胖的流行率增加了两倍,根据世界卫生组织的数据,2016年全球超过6.5亿18岁及以上的成年人肥胖。过量脂肪组织是促炎症因子的主要来源,可导致慢性全身炎症和一系列代谢紊乱,包括2型糖尿病(T2DM)[6]。 严重的Covid-19感染和细胞因子风暴在BMI升高和糖尿病的患者中更为明显[3,7],这表明肥胖相关的炎症和代谢功能障碍加剧了疾病的严重程度,从而导致两种全球大流行的交叉 。
目前,一些新冠疫苗已获批用于接种。然而,实现群体免疫仍存在一定的障碍,例如对疫苗顾虑、制造延迟、分布差异以及SARS-CoV-2变异等因素增加了新冠肺炎持续爆发的可能性[8]。此外,已有研究显示肥胖人群对疫苗接种反应不佳[9,10]。 不健康的代谢状况,包括促炎细胞因子水平和血糖的升高,与对Covid-19的免疫反应失调显著相关 [11,12]。
高血糖与Covid-19感染潜在机制:与葡萄糖代谢激活途径相关
血糖控制不良与Covid-19的严重程度独立相关,并且死亡率风险显著增加[13-15、16、17]。这种关联的病理生理机制可能与增加的葡萄糖代谢途径激活有关。病毒感染引起的六糖胺生物合成的增加,通过干扰素调节因子-5(IRF5)的上调[18,19],进而导致细胞炎症因子风暴,其中IRF-5是肥胖个体脂肪组织中升高的代谢性炎症的标志物[20-21]。 高血糖与肥胖糖尿病患者单核细胞中SARS-CoV-2复制增加有关,病毒的持续增殖,诱导细胞因子风暴,导致T细胞功能障碍 [7]。此外, 高血糖症可增强SARS-CoV-2受体血管紧张素转换酶2(ACE2)的糖基化[22,23],糖基化ACE2而不是总ACE2的表达与肺中的病毒结合和融合相关 [24]。

图1 高血糖介导Covid-19发病机制
*注:ARDS,急性呼吸窘迫综合征;HIF-1α,缺氧诱导因子-1α;ROS,活性氧。
肥胖、免疫与Covid-19:组织微环境从抗炎到促炎表型转变
肥胖所致的脂肪积累使组织微环境从抗炎到促炎表型转变,主要表现为促炎(类M1)与抗炎(类M2)巨噬细胞的比率增加,辅助T1细胞(Th1)向Th17和CD8+细胞毒性T细胞极化,调节性T细胞减少[6]。
这些变化使炎症细胞因子水平升高,例如,白细胞介素-6(IL-6)、肿瘤坏死因子(TNFα)、IL-1β、瘦素[6,25]、单核细胞数量增加。促炎细胞因子的增加可能会加剧病毒诱导的细胞因子风暴,驱动组织损伤,导致急性呼吸窘迫综合征(ARDS)、多器官衰竭,最终导致死亡[11,26]。 肥胖人群中免疫细胞激活减弱和记忆性T细胞反应的受损会导致病毒感染的预后不良,更为严重的是降低了这部分人群对于疫苗的反应性 [9]。肥胖会导致肠道生理受损,包括屏障渗透性的增加和微生物功能失调[27]。由于胃肠道系统(GI)被认为是SARS-CoV-2进入的靶点,肠道健康状况不佳可能是Covid-19发展的一个诱发因素。
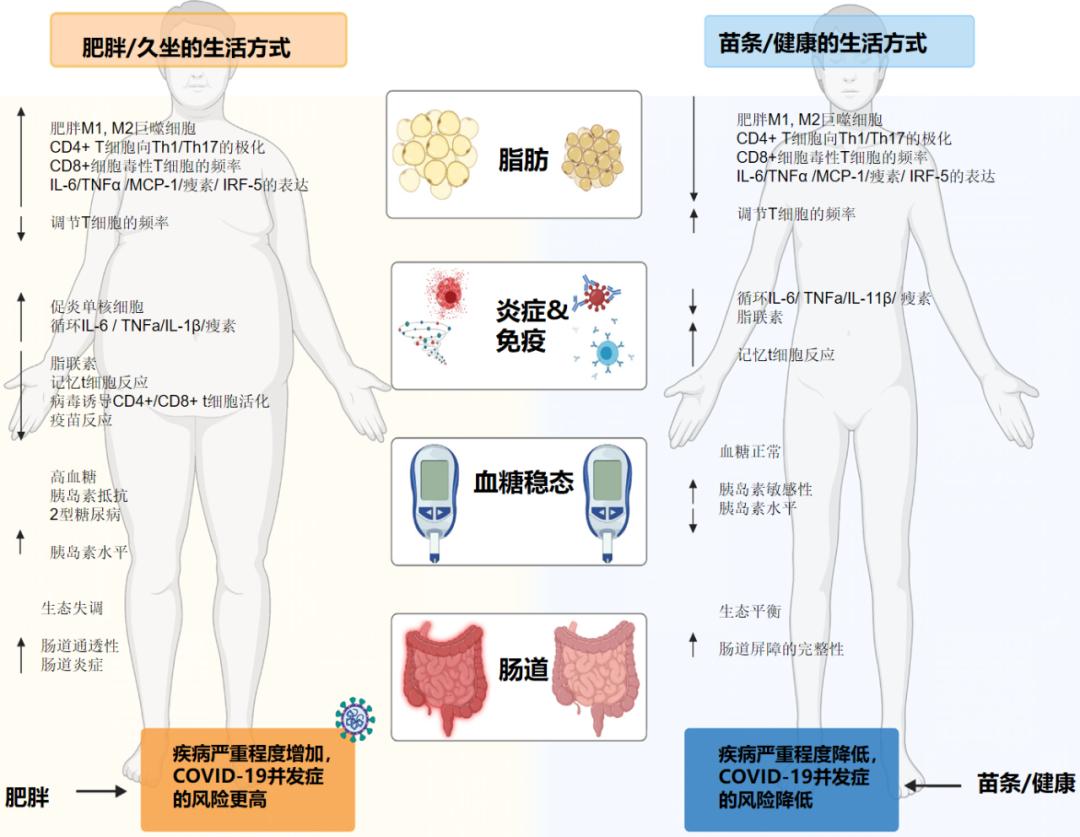
图2 肥胖对代谢和免疫的影响
轻断食会是Covid-19流行期间的突破口吗?
综上所述,体重增加引起的病理生理变化导致代谢功能障碍、慢性炎症、免疫反应受损和多系统疾病,增加了Covid-19严重性。这时候比以往任何时候都更需要生活方式的改变,促进代谢和免疫健康,以减轻Covid-19的影响。为了解决传统、限制热量等方面的减重阻碍,时下流行的间歇性禁食(IF)已经作为一种实用而有效的替代方案越来越受欢迎。
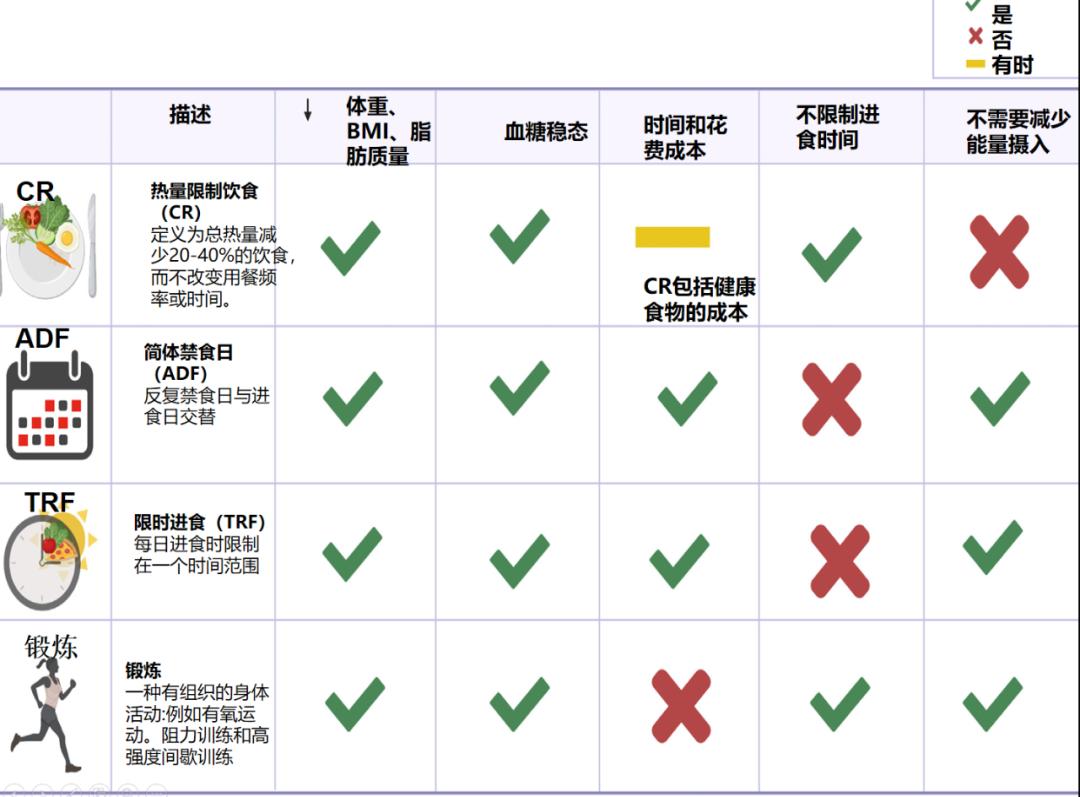
图3 健康生活方式比较
*注:CR:热量限制性饮食,定义为总热量减少20-40%的饮食,而不改变用餐频率或时间;ADF:IF的一种,反复禁食日与进食日交替;TRF:IF的一种,每日进食时限制在一个时间范围。
最近,在许多临床前和临床研究中,IF已被证实是减重和控制血糖的有效干预措施[28-30,31,32]。虽然中强度到高强度的定期锻炼可以减重减脂;然而,有证据表明,仅靠运动带来的体重减少微乎其微[33]。一项荟萃分析发现[34],与单独的阻力训练相比,IF与阻力训练运动联合对BMI和脂肪质量的减少有一些影响,这表明需要进一步研究来验证运动和IF相结合的优势。
正如最近的证据表明,体育活动可以预防Covid-19严重的后果一样[35],体育活动和饮食干预的生活方式很可能是减轻Covid-19影响的理想方法。然而,在Covid-19大流行期间,社交距离成了参与体育活动的障碍,而IF的优势在于它是一种零成本的时间管理方法,而锻炼通常需要时间、资源和设施配合同时需要克服不活动的相关障碍[36]。因此,虽然锻炼和IF结合是体重管理富有吸引力的选择,但IF对一些人来说可能是一种更实用的方法,尤其是在全球大流行期间。
IF的多方面健康获益,新冠肺炎下如何选择健康饮食?
这篇综述分析了动物实验及人类的研究,阐述了IF对脂肪、代谢健康、葡萄糖稳态、抗病毒免疫反应、长期免疫、炎症标记物和肠道的获益,这里不作阐述。 值得关注的仍然是围绕着Covid-19疫苗接种对肥胖人群的长期有效性,因为在这部分人群中记忆T细胞中的激活和对疫苗接种的反应迟钝 [37]。在最近的一项研究中,小鼠肥胖相关微环境促进了更具毒性的致病性流感病毒毒株的出现,这导致瘦型宿主的疾病严重程度更大[38]。这或可带来有趣的可能性,即 肥胖代谢表型可能影响新一代Covid-19突变体 。
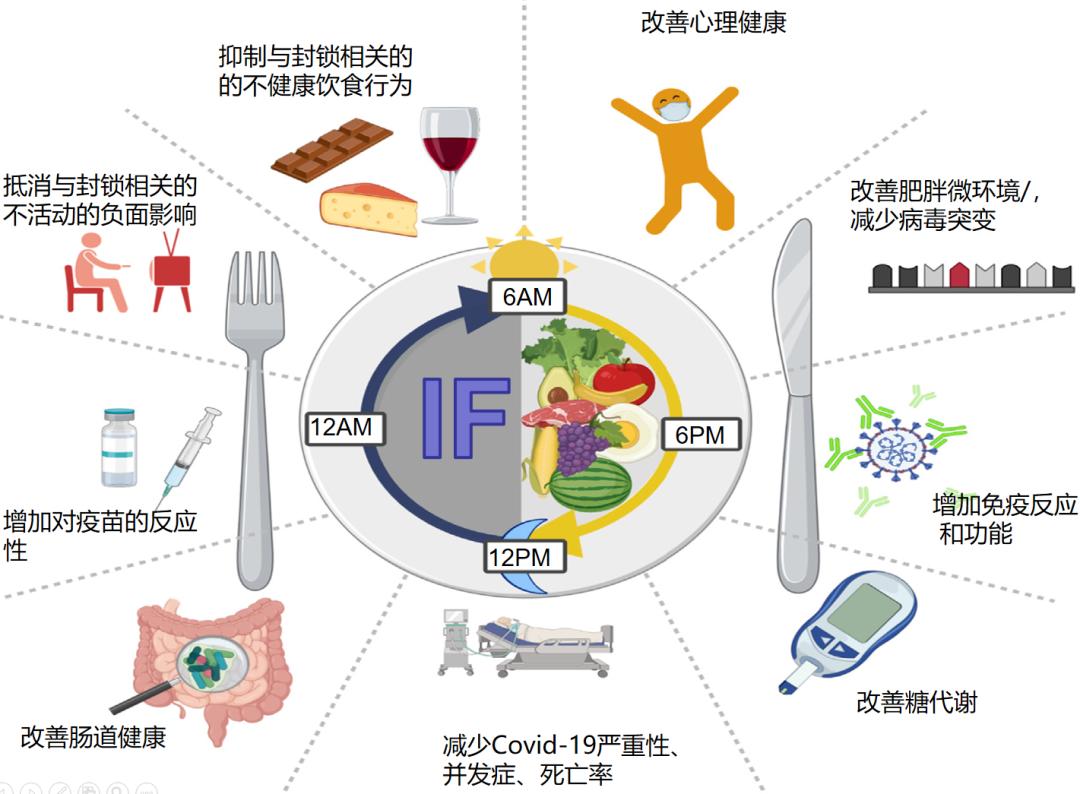
图4 Covid-19流行期间的IF
由于Covid-19大流行,数十亿人继续采取各种隔离措施,甚至实施*锁封**政策。这导致了久坐不动的生活方式的增加和长期的社会孤立,这对身心健康均有不利影响[39,40]。在这场大流行期间饮食习惯发生了巨大的变化,主要表现为更多的在家做饭和吃饭时间,更少的在家锻炼,无聊和疫情压力都会导致暴饮暴食和盲目的零食增加。
这些改变也引起了人们对健康饮食的关注,寻求提高免疫,有益健康的策略[41,42]。鉴于目前的体育活动的机会有限,饮食改变,IF治疗方案可能是一种实际的方式,特别是TRF方案可抑制一天的饥饿感,减少非饥饿零食摄入和盲目地进食[43]。此外,IF也与情绪和健康的改善有关[44,45]。
TRF或IF可能比传统饮食有更高的可持续性,因为它允许个人在较短的时间内保持他们的日常饮食。在许多研究中,减少每天的进食时间会导致热量摄入的无意减少,从而导致体重减轻和相关的代谢益处[46]。虽然IF已被证实有积极的代谢获益,如改善葡萄糖稳态,即便在没有减轻体重和/或整体热量消耗没有变化的情况下[31,32,43]。必须注意的是,这些发现的大多数证据仅来自小型对照临床试验。
更为重要的是,IF作为一种治疗方案尚未涉及Covid-19感染,可能在新冠疫情下,IF和锻炼的结合才是最有效的方式。最后,还有一些人担心,IF策略是否适合每个人,减少热量摄入的饮食方案可能会利用免疫抑制作用,削弱抗SARS-CoV-2的免疫反应[47], 因此对于免疫力减弱的人群中,还需谨慎 。此外,IF对老年人口的影响的报告也很有限,虽然已经有研究证实IF在老年人口中的身心获益(>65岁)[48],但在老年人中,实施IF必须谨慎行事。
总 结
综上所述,虽然IF是一个令人兴奋和日益被关注的研究领域,但尚需进一步的研究来确定IF对抗病毒免疫的影响,特别是在Covid-19的背景下,鉴于IF在改善代谢健康方面表现出了深远影响,现在采用这种健康的生活方式策略是比以往任何时候都更好的时机。
文/晨少
责编/Jane
参考文献
1.Kafi N. Ealey, Joy Phillips, COVID-19 and obesity: fighting two pandemics with intermittent fasting Hoon-Ki Sung.Trends in Endocrinology & Metabolism, Month 2021, Vol. xx, No. xx https://doi.org/10.1016/j.tem.2021.06.004 1
2.Gao, M. et al. (2021) Associations between body-mass index and COVID-19 severity in 6.9 million people in England: a prospective, community-based, cohort study. Lancet Diabetes Endocrinol. 9, 350–359
3.Kang, Z. et al. (2020) Obesity is a potential risk factor contributing to clinical manifestations of COVID-19. Int. J. Obes.(Lond.) 44, 2479–2485
4.Simonnet, A. et al. (2020) High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation. Obesity (Silver Spring) 28, 1195–1199
5.O’Hearn, M. et al. (2021) Coronavirus disease 2019 hospitalizations attributable to cardiometabolic conditions in theUnited States: a comparative risk assessment analysis.J. Am. Heart Assoc. 10, e019259
6.Alarcon, P.C. et al. (2021) Adipocyte inflammation and pathogenesis of viral pneumonias: an overlooked contribution. Mucosal Immunol. Published online May 6, 2021. https://doi.org/
10.1038/s41385-021-00404-8
7.Codo, A.C. et al. (2020) Elevated glucose levels favor SARSCoV-2 infection and monocyte response through a HIF-1α/glycolysis-dependent axis. Cell Metab. 32, 437–446.e435
8.Aschwanden, C. (2021) Five reasons why COVID herd immunity is probably impossible. Nature 591, 520–522
9.Neidich, S.D. et al. (2017) Increased risk of influenza among vaccinated adults who are obese. Int. J. Obes. (Lond.) 41, 1324–1330
10.Paich, H.A. et al. (2013) Overweight and obese adult humans have a defective cellular immune response to pandemic H1N1 influenza A virus. Obesity (Silver Spring) 21, 2377–2386
11.Del Valle, D.M. et al. (2020) An inflammatory cytokine signature predicts COVID-19 severity and survival. Nat. Med. 26, 1636–1643
12.Laguna-Goya, R. et al. (2020) IL-6-based mortality risk model for hospitalized patients with COVID-19. J. Allergy Clin.Immunol. 146, 799–807.e799
13.Bode, B. et al. (2020) Glycemic characteristics and clinical outcomes of COVID-19 patients hospitalized in the United States.J. Diabetes Sci. Technol. 14, 813–821
14.Zhu, L. et al. (2020) Association of blood glucose control andoutcomes in patients with COVID-19 and pre-existing type 2 diabetes. Cell Metab. 31, 1068–1077.e1063
15.Smith, S.M. et al. (2021) Impaired glucose metabolism in patients with diabetes, prediabetes, and obesity is associated with severe COVID-19. J. Med. Virol. 93, 409–415
16.Iacobellis, G. et al. (2020) Admission hyperglycemia and radiological findings of SARS-CoV2 in patients with and without diabetes. Diabetes Res. Clin. Pract. 164, 108185
17.Wang, S. et al. (2020) Fasting blood glucose at admission is an independent predictor for 28-day mortality in patients with COVID-19 without previous diagnosis of diabetes: a multicentre retrospective study. Diabetologia 63, 2102–2111
18.Wang, Q. et al. (2020) O-GlcNAc transferase promotes influenza A virus-induced cytokine storm by targeting interferon regulatory factor-5. Sci. Adv. 6, eaaz7086
19.Wang, X. et al. (2018) Expression levels of interferon regulatory factor 5 (IRF5) and related inflammatory cytokines associated with severity, prognosis, and causative pathogen in patients
with community-acquired pneumonia. Med. Sci. Monit. 24, 3620–3630
20.Sindhu, S. et al. (2020) Enhanced adipose expression of interferon regulatory factor (IRF)-5 associates with the signatures of metabolic inflammation in diabetic obese patients. Cells 9, 730
21.Dalmas, E. et al. (2015) Irf5 deficiency in macrophages promotes beneficial adipose tissue expansion and insulin sensitivity during obesity. Nat. Med. 21, 610–618
22.Walls, A.C. et al. (2020) Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell 181, 281–292.e286
23.Mehdipour, A.R. and Hummer, G. (2021) Dual nature of human ACE2 glycosylation in binding to SARS-CoV-2 spike. Proc. Natl Acad. Sci. U. S. A. 118, e2100425118
24.Brufsky, A. (2020) Hyperglycemia, hydroxychloroquine, and the COVID-19 pandemic. J. Med. Virol. 92, 770–775
25.Mohamed-Ali, V. et al. (1997) Subcutaneous adipose tissue releases interleukin-6, but not tumor necrosis factor-alpha, in vivo. J. Clin. Endocrinol. Metab. 82, 4196–4200
26.Zhang, D. et al. (2021) Frontline science: COVID-19 infection induces readily detectable morphologic and inflammationrelated phenotypic changes in peripheral blood monocytes.J. Leukoc. Biol. 109, 13–22
27.Yeoh, Y.K. et al. (2021) Gut microbiota composition reflects disease severity and dysfunctional immune responses in patients with COVID-19. Gut 70, 608–706
28.Gabel, K. et al. (2018) Effects of 8-hour time restricted feeding on body weight and metabolic disease risk factors in obese adults: a pilot study. Nutr. Healthy Aging 4, 345–353
29.Kesztyus, D. et al. (2019) Adherence to time-restricted feeding and impact on abdominal obesity in primary care patients: results of a pilot study in a pre-post design.Nutrients 11, 2854
30.Cienfuegos, S. et al. (2020) Effects of 4- and 6-h time-restricted feeding on weight and cardiometabolic health: a randomized controlled trial in adults with obesity. Cell Metab. 32,
366–378.e363
31.Kim, Y.H. et al. (2019) Thermogenesis-independent metabolic benefits conferred by isocaloric intermittent fasting in ob/ob mice. Sci. Rep. 9, 2479
32.Kim, K.H. et al. (2017) Intermittent fasting promotes adipose thermogenesis and metabolic homeostasis via VEGFmediated alternative activation of macrophage. Cell Res. 27,1309–1326
33.Bellicha, A. et al. (2021) Effect of exercise training on weight loss, body composition changes, and weight maintenance in adults with overweight or obesity: an overview of 12 systematic reviews and 149 studies. Obes. Rev. Published online May 6, 2021. https://doi.org/10.1111/obr.13256
34.Ashtary-Larky, D. et al. (2021) Effects of intermittent fasting combined with resistance training on body composition: a systematic review and meta-analysis. Physiol. Behav. 237,
113453
35.Sallis, R. et al. (2021) Physical inactivity is associated with a higher risk for severe COVID-19 outcomes: a study in 48 440 adult patients. Br. J. Sports Med. Published online April 13, 2021. https://doi.org/10.1136/bjsports-2021-104080
36.Herazo-Beltran, Y. et al. (2017) Predictors of perceived barriers to physical activity in the general adult population: a crosssectional study. Braz. J. Phys. Ther. 21, 44–50
37.Pellini, R. et al. (2021) Obesity may hamper SARS-CoV-2- specific vaccine. medRxiv Published online February 26, 2021. https://doi.org/10.1101/2021.02.24.21251664
38.Honce, R. et al. (2020) Obesity-related microenvironment promotes emergence of virulent influenza virus strains. mBio 11, e03341-19
39.Pietrabissa, G. and Simpson, S.G. (2020) Psychological consequences of social isolation during COVID-19 outbreak.Front. Psychol. 11, 2201
40.Stockwell, S. et al. (2021) Changes in physical activity and sedentary behaviours from before to during the COVID-19 pandemic lockdown: a systematic review. BMJ Open Sport Exerc. Med. 7, e000960
41.Di Renzo, L. et al. (2020) Eating habits and lifestyle changes during COVID-19 lockdown: an Italian survey. J. Transl. Med. 18, 229
42.Ruiz-Roso, M.B. et al. (2020) COVID-19 lockdown and changes of the dietary pattern and physical activity habits in a cohort of patients with type 2 diabetes mellitus. Nutrients 12, 2327
43.Sutton, E.F. et al. (2018) Early time-restricted feeding improves insulin sensitivity, blood pressure, and oxidative stress even without weight loss in men with prediabetes. Cell Metab. 27,
1212–1221.e1213
44.Hussin, N.M. et al. (2013) Efficacy of fasting and calorie restriction (FCR) on mood and depression among ageing men.J. Nutr. Health Aging 17, 674–680.
45.Igwe, O. et al. (2020) A review of effects of calorie restriction and fasting with potential relevance to depression.Prog. Neuropsychopharmacol. Biol. Psychiatry. Published online December 11, 2020. https://doi.org/10.1016/j.pnpbp.2020.110206
46.Gill, S. and Panda, S. (2015) A smartphone app reveals erratic diurnal eating patterns in humans that can be modulated for health benefits. Cell Metab. 22, 789–798
47.Duriancik, D.M. et al. (2018) Age, calorie restriction, and age of calorie restriction onset reduce maturation of natural killer cells in C57Bl/6 mice. Nutr. Res. 55, 81–93
48.Anton, S.D. et al. (2019) The effects of time restricted feeding on overweight, older adults: a pilot study. Nutrients 11, 1500