
中国专家首次采用益生菌基因改造后能分泌独有的GLP-1类似肽,通过胃肠给予菌群达到显著降低体重、抑制体重增加,降低体内脂肪垫的重量。还能降低肝脏甘油三酯、总胆固醇的含量,改善肝脏组织的病理变化,对肥胖小鼠肝脏具有一定的保护作用。这意味着有望通过改变肠道菌群分泌功能,达到替代药物改善代谢异常疾病的潜在可能。

该研究由战略支援部队特*医色**学中心顾建文教授和利通生物医药科技马婕博士等主持,研究成果发表在2020年1月23日,OBESITY 杂志。
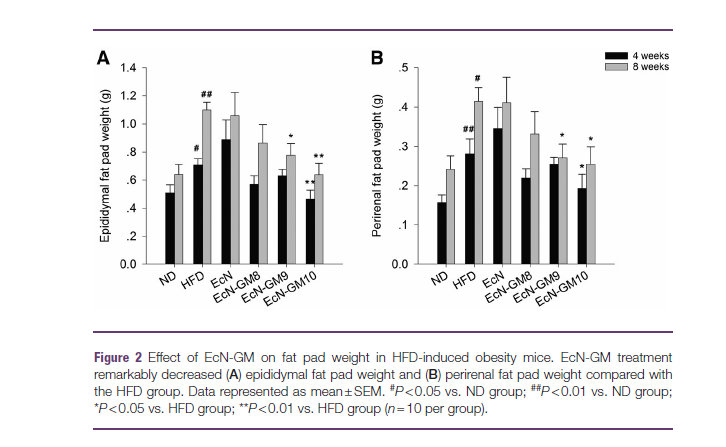
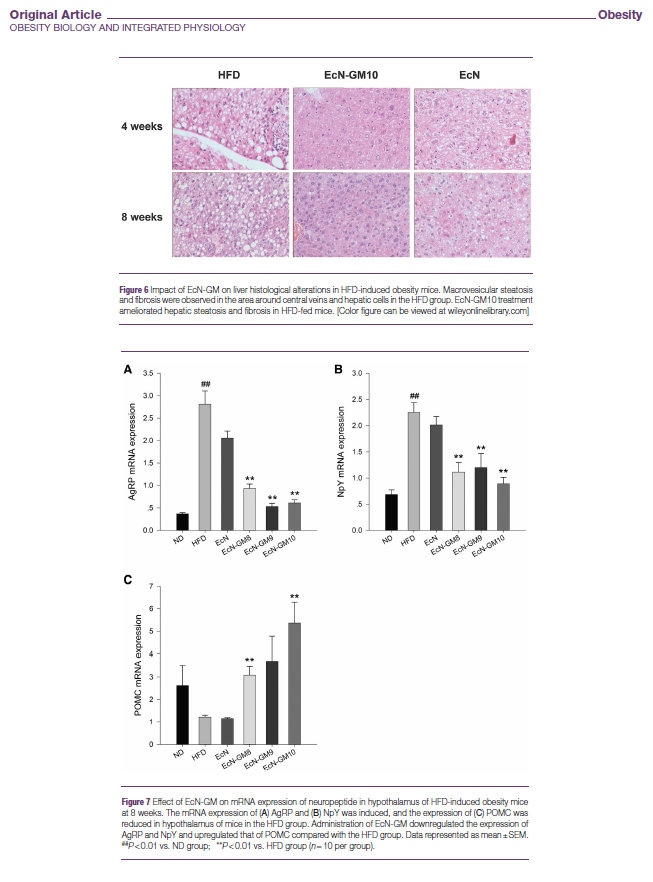
在前期研究中,通过GLP-1原始序列的改造,该团队选取其中最为有潜力的序列构建工程菌株表达,基因工程大肠杆菌Nissle1917。研究的主要目的是初步探讨工程菌EcN-GM是否可作为治疗肥胖的新方法。本研究的结果表明,在高脂饮食诱导的肥胖小鼠中,连续灌胃8周EcN-GM可以显著降低体重、抑制体重增加,降低体内脂肪垫的重量。上述结果与实验中观察到的小鼠摄食量显著减少的趋势是平行的。这一结果与其他研究显示GLP-1和类似物GLP-1可通过抑制啮齿动物和小型猪的食欲来减轻体重是一致的(24-26)。同时,EcN-GM治疗8周后,可降低小鼠血糖水平。EcN-GM还能降低肝脏甘油三酯、总胆固醇的含量,改善肝脏组织的病理变化,对肥胖小鼠肝脏具有一定的保护作用。
综上所述,顾建文团队研究首次揭示了基因工程益生菌(EcN-GM)对肥胖、血糖和肝脂肪变性的有益作用。这可能与调节下丘脑能量摄入和消耗的神经肽表达有关。本研究成功地证明了工程菌EcN-GM可作为一种具有潜力的、新的体重管理方法。

肥胖通常是指的异常或过量脂肪累积,近年来已成为世界范围内目前最重要的公共卫生问题之一(1)。肥胖为多种慢性疾病的主要诱因,如2型糖尿病、高血压、心血管疾病、非酒精脂肪肝(NAFLD)、中风和骨关节炎等(2 - 5),给家庭和社会带来了巨大的经济负担(6、7)。
有研究表明,生活方式如饮食干预或运动等行为是预防和改善肥胖的主要方法,但是大部分人无法长期坚持(8)。如果生活方式干预无法达到理想的效果,则需要考虑采取药物治疗的方法控制肥胖。多年来,多种治疗肥胖的药物由于其严重的副作用,已经退出市场(9)。因此,肥胖症的治疗急需新的方法和策略。

近来来,肠道分泌的内源性肽类在能量稳态中的重要作用引起了研究者们的关注(10)。胰高血糖素样肽-1 (GLP-1)是由30个氨基酸组成,其对葡萄糖代谢和体重的影响越来越被人们所重视(11)。进食刺激后,肠道内分泌细胞分泌GLP-1。GLP-1可呈葡萄糖浓度依赖性刺激胰岛素分泌,降低血浆胰高血糖素水平,并可直接作用于β细胞促进其增殖与分化,从而降低体重,避免低血糖风险 (12)。但是由于二肽基肽酶-4 (DPP-4)的快速降解,循环中的GLP-1的半衰期很短,只有2-5分钟(11,13)。因此,本课题组对GLP-1原始序列进行了一系列改造,构建了新的GLP-1(GM)序列。
与此同时,应用GLP-1和GLP-1类似物所面临的最大挑战在于多肽类药物只能通过注射的方式给药,用药顺应性差,不适合肥胖症等慢*病治性疗**的长期给药,需要开发更新颖的给药方法(14,15)。大肠杆菌Nissle1917 (E. coli Nissle1917, EcN)不携带任何致病基因,对人体没有致病性,通常作为益生菌用于治疗肠道感染和疾病(16)。近年来,多项研究显示,EcN进行基因工程改造后可作为外源性治疗因子的载体用于肥胖症、糖尿病、肠道疾病等疾病的治疗(17-19)。

由于患者的依从性,口服给药仍然是治疗慢性疾病的首选给药途径。因此,该方法也可作为一种新的口服给药的活菌治疗方式,将生物活性分子递送至肠道内,以避免降解。经过基因改造我们构建了EcN-GM菌株,本研究在高脂饮食诱导的肥胖小鼠中对EcN-GM改善肥胖的作用进行了初步评价。
下一步将在应用到人体作用方面还需大量的进一步研究。有望开拓生物类似药物研究。


参考文献
1. NufferW, Trujillo JM, MegyeriJ. A Comparison of New Pharmacological Agents for the Treatment of Obesity. Ann Pharmacother 2016;50: 376-388.
2. Emerging Risk Factors C, Wormser D, KaptogeS, DiAngelantonioE, Wood AM, Pennells L, et al. Separate and combined associations of body-mass index and abdominal adiposity with cardiovascular disease: collaborative analysis of 58 prospective studies. Lancet 2011;377: 1085-1095.
3. Singh GM, Danaei G, Farzadfar F, Stevens GA, Woodward M, Wormser D, et al. The age-specific quantitative effects of metabolic risk factors on cardiovascular diseases and diabetes: a pooled analysis. PLoS One 2013;8: e65174.
4. KirpichIA, Marsano LS, McClainCJ. Gut-liver axis, nutrition, and non-alcoholic fatty liver disease. Clin Biochem 2015;48: 923-930.
5. ApovianCM, AronneLJ, Bessesen DH, McDonnell ME, Murad MH, PagottoU, et al. Pharmacological management of obesity: an endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2015;100: 342-362.
6. Collaborators GBDO, Afshin A, Forouzanfar MH, Reitsma MB, Sur P, Estep K, et al. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N EnglJ Med 2017;377:13-27.
7. Ng M, Fleming T, Robinson M, Thomson B, Graetz N, Margono C, et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 2014;384: 766-781.
8. Rankin W, Wittert G. Anti-obesity drugs. Curr Opin Lipidol 2015;26: 536-543.
9. KangJG, Park CY. Anti-Obesity Drugs: A Review about Their Effects and Safety. Diabetes Metab J 2012;36: 13-25.
10. Zhao W, Wang L, Chen P, Zhao J, Qi Y, Chi S, et al. 7-aa peptide mimic from HVR1 of HCV protects hepatic injury in rats by reduced expression of key pro-inflammatory factors. Inflamm Allergy Drug Targets 2010;9: 135-145.
11. HolstJJ. The physiology of glucagon-like peptide 1. Physiol Rev 2007;87: 1409-1439.
12. NauckMA, Meier JJ. Glucagon-like peptide 1 and its derivatives in the treatment of diabetes. Regul Pept 2005;128: 135-148.
13. NeffLM, Kushner RF. Emerging role of GLP-1 receptor agonists in the treatment of obesity. Diabetes Metab Syndr Obes 2010;3: 263-273.
14. DuanF, Curtis KL, March JC. Secretion of insulinotropic proteins by commensal bacteria: rewiring the gut to treat diabetes. Appl Environ Microbiol 2008;74: 7437-7438.
15. DuanFF, LiuJH, March JC. Engineered commensal bacteria reprogram intestinal cells into glucose-responsive insulin-secreting cells for the treatment of diabetes. Diabetes 2015;64: 1794-1803.
16. Hancock V, Dahl M, Klemm P. Probiotic Escherichia coli strain Nissle 1917 outcompetes intestinal pathogens during biofilm formation. J Med Microbiol 2010;59: 392-399.
17. Chen Z, Guo L, Zhang Y, Walzem RL, Pendergast JS, Printz RL, et al. Incorporation of therapeutically modified bacteria into gut microbiota inhibits obesity. J Clin Invest 2014;124: 3391-3406.
18. Hendrikx T, Duan Y, Wang Y, Oh JH, Alexander LM, Huang W, et al. Bacteria engineered to produce IL-22 in intestine induce expression of REG3G to reduce ethanol-induced liver disease in mice. Gut 2018.
19. Isabella VM, Ha BN, Castillo MJ, Lubkowicz DJ, Rowe SE, Millet YA, et al. Development of a synthetic live bacterial therapeutic for the human metabolic disease phenylketonuria. Nat Biotechnol 2018;36: 857-864.
20. Bray GA, Fruhbeck G, Ryan DH, Wilding JP. Management of obesity. Lancet 2016;387: 1947-1956.
21. Wei P, Yang Y, Li T, Ding Q, SunH. A engineered Bifidobacterium longum secreting a bioative penetratin-Glucagon-like peptide 1 fusion protein enhances Glucagon-like peptide 1 absorption in the intestine. Journal of microbiology and biotechnology 2015.
22. Chen T, Tian P, Huang Z, Zhao X, Wang H, Xia C, et al. Engineered commensal bacteria prevent systemic inflammation-induced memory impairment and amyloidogenesis via producing GLP-1. Applied microbiology and biotechnology 2018;102: 7565-7575.
23. Zeng Z, Yu R, Zuo F, Zhang B, Peng D, Ma H, et al. Heterologous Expression and Delivery of Biologically Active Exendin-4 by Lactobacillus paracasei L14. PloS one 2016;11: e0165130.
24. RaunK, von Voss P, KnudsenLB.Liraglutide, a once-daily human glucagon-like peptide-1 analog, minimizes food intake in severely obese minipigs. Obesity 2007;15: 1710-1716.
25. Hansen HH, Hansen G, Paulsen S, Vrang N, Mark M, Jelsing J, et al. The DPP-IV inhibitor linagliptin and GLP-1 induce synergistic effects on body weight loss and appetite suppression in the diet-induced obese rat. Eur J Pharmacol 2014;741: 254-263.
26. Raun K, von Voss P, Gotfredsen CF, Golozoubova V, Rolin B, Knudsen LB. Liraglutide, a long-acting glucagon-like peptide-1 analog, reduces body weight and food intake in obese candy-fed rats, whereas a dipeptidyl peptidase-IV inhibitor, vildagliptin, does not. Diabetes 2007;56: 8-15.
27. Kazaks A, Stern JS. Obesity: food intake. Prim Care 2003;30: 301-316, vi.
28. YadavH, Lee JH, Lloyd J, Walter P, Rane SG. Beneficial metabolic effects of a probiotic via butyrate-induced GLP-1 hormone secretion. J Biol Chem 2013;288: 25088-25097.
29. SohnJW. Network of hypothalamic neurons that control appetite. BMB reports 2015;48: 229-233.
30. TodaC, SantoroA, KimJD, DianoS.POMC Neurons: From Birth to Death. Annu Rev Physiol 2017;79: 209-236.
31. Jennings JH, Ung RL, Resendez SL, Stamatakis AM, Taylor JG, Huang J, et al. Visualizing hypothalamic network dynamics for appetitive and consummatory behaviors. Cell 2015;160: 516-527.
32. Krashes MJ, Shah BP, Madara JC, Olson DP, Strochlic DE, Garfield AS, et al. An excitatory paraventricular nucleus to AgRP neuron circuit that drives hunger. Nature 2014;507: 238-242.
33. Cooke D, Bloom S. The obesity pipeline: current strategies in the development of anti-obesity drugs. Nature reviews Drug discovery 2006;5: 919-931.
34. DianoS. New aspects of melanocortin signaling: a role for PRCP in alpha-MSH degradation. Front Neuroendocrinol 2011;32: 70-83.
35. KanoskiSE, Hayes MR, Skibicka KP. GLP-1 and weight loss: unraveling the diverse neural circuitry. Am J PhysiolRegulIntegr Comp Physiol 2016;310: R885-895.