编者按:20年前,根据两项大型III期研究,伊马替尼获得批准,并彻底改变了晚期GIST的治疗。此后,二线、三线和四线 TKI(如舒尼替尼、瑞戈非尼和利普替尼)已被美国食品药品监督管理局(FDA)批准用于治疗晚期GIST。在本文中,来自南澳大利亚伊丽莎白女王医院和阿德莱德肿瘤学和血液学(Genesis Care)肿瘤内科医生Tiffany Foo,弗林德斯癌症创新中心的肿瘤内科医生,澳大利亚弗林德斯大学医学与公共卫生学院的副教授Amitesh C. Roy以及澳大利亚莫纳什大学公共卫生与预防医学学院癌症研究项目的负责人John Zalcberg博士盘点了晚期不可切除的胃肠道间质瘤靶向治疗的现状及未来研究方向。
酪氨酸激酶*制剂抑**(TKI)改变了我们管理许多恶性肿瘤的方式,包括胃肠道间质瘤(GIST)。约20年前,一项II期研究的结果表明,伊马替尼在大约一半晚期GIST患者中产生了持续的客观缓解[1]。根据两项大型III期研究,伊马替尼获得批准,并彻底改变了晚期GIST的治疗[2,3]。此后,二线、三线和四线 TKI(如舒尼替尼、瑞戈非尼和利普替尼)已被美国食品药品监督管理局(FDA)批准用于治疗晚期GIST。此外,阿伐普替尼已被批准用于具有特定GIST亚群的患者,该亚群携带PDGFRA基因外显子18突变,包括D842V突变[4]。

Tiffany Foo博士
分子评估在晚期、不可切除GIST中的重要性
用于治疗GIST的TKI的出现支持对所有新诊断GIST患者进行常规分子检测的需求。不可切除的晚期GIST的分子改变可预测对伊马替尼的敏感性,并具有治疗和预后意义。约80%的GIST在原癌基因KIT中具有功能获得突变作为驱动事件[13,14]。KIT中最常见的突变发生在外显子11,其次是外显子9。伊马替尼是外显子11或外显子9中携带敏感KIT突变的晚期GIST患者的标准治疗。然而,来自EORTC-62005和SWOG-S0033试验的证据以及胃肠道基质肿瘤荟萃分析组的荟萃分析表明,携带KIT外显子9突变的晚期GIST患者使用较高剂量的伊马替尼(每日800 mg)可改善无进展生存期 (PFS)[5,7,15],这被认为是由于外显子9编码KIT的细胞外结构域,该GIST亚组中的细胞内激酶结构域与野生型KIT相似,导致对伊马替尼的固有耐药性[16]。
在激活性KIT改变之后,GIST中第二常见的遗传驱动事件是PDGFRA的致病性改变[17,18],携带这种突变的GIST对伊马替尼具有固有的耐药性。2019 年,I期NAVIGATOR试验证实,阿伐普替尼在以PDGFRA D842V 突变为特征的GIST患者中具有临床活性,还观察到总缓解率为88%和9%的完全缓解率[12]。这项具有里程碑意义的试验导致FDA批准阿瓦普替尼用于治疗PDGFRA基因外显子18突变的晚期GIST患者 ,其中最常见的是D842V突变[4]。随后,VOYAGER研究进一步证实,与瑞戈非尼相比,PDGFRA D842V突变GIST患者使用阿瓦普替尼具有较高的反应率和更长的PFS。本研究进一步强调了GIST患者分子检测的重要性,以帮助选择最合适的治疗方法。

John Zalcberg博士
如何克服对伊马替尼的继发性耐药性?
尽管最初对伊马替尼的反应率很高,但疾病进展通常发生在治疗后 24 个月内,对伊马替尼的继发性耐药仍然是一个挑战。虽然对伊马替尼的原发性耐药性通常是由伊马替尼固有耐药性突变引起的(例如,PDGFRA D842V突变的GIST),但继发性耐药主要是由于携带继发性KIT突变的耐药亚克隆的出现[23]。这种继发突变之间存在显著的异质性,这使得开发单一有效的治疗药物具有挑战性。尽管舒尼替尼和瑞戈非尼在表现出其中一些突变的GIST患者中均有活性,但其活性有限。随后,出现其他耐药亚克隆使得二线和三线药物治疗与中位PFS短相关(舒尼替尼为5.6个月,瑞戈非尼为4.8个月)[8,10]。
此外,迫切需要新的治疗策略来克服这些继发突变,以确保在伊马替尼失败后获得更持久的反应。利普替尼是一种新型开关控制TKI,通过结合开关口袋和激活环,将激酶锁定在非活性状态,这抑制了KIT和PDGFRA激酶信号通路。2020年,根据INVICTUS试验的结果,利普替尼被FDA批准用于接受3线或更多线激酶*制剂抑**的患者[26]。在这项3期随机安慰剂对照试验中,与安慰剂相比,接受利普替尼治疗的受试者在中位总生存期方面具有临床意义和统计学意义的改善(尽管随后与利普替尼组交叉)[11]。
在INVICTUS研究取得有希望的结果之后,INTRIGUE研究在二线治疗中比较了利普替尼与舒尼替尼[27]。这项国际III期研究未达到其主要终点。尽管利普替尼显示出有意义的临床活性,但与舒尼替尼相比,PFS没有统计学上的显著改善[28]。利普替尼组和舒尼替尼组治疗人群的中位PFS分别为8.0个月和8.3个月(HR 1.05,95% CI [0.82,1.33]; P=0.72)。尽管INTRIGUE研究无法确定等效性,但利普替尼的PFS与舒尼替尼相当。
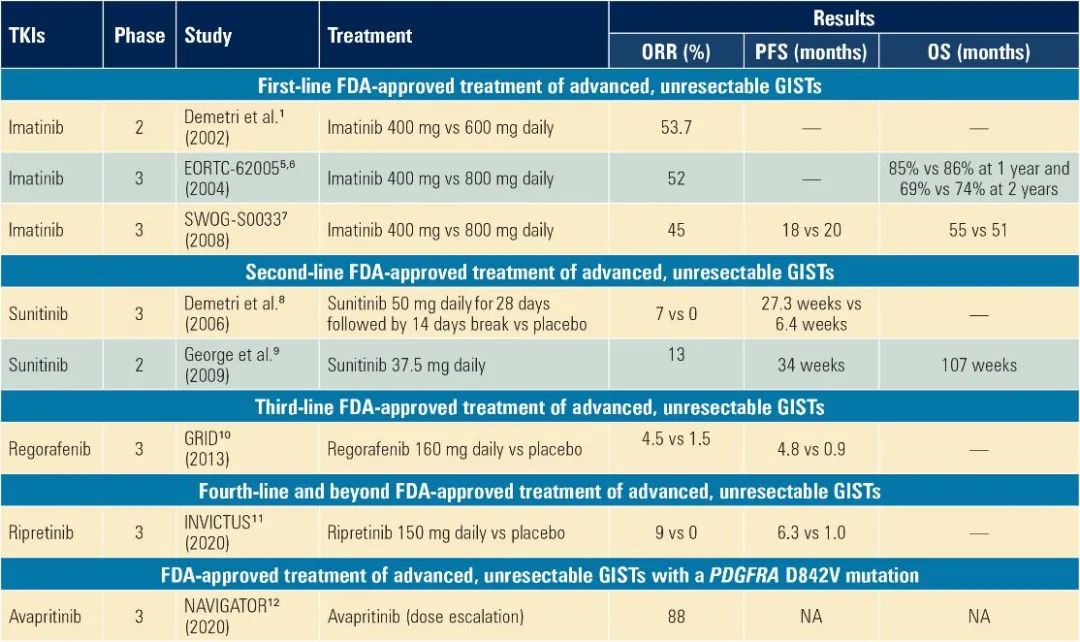
图1 已获FDA批准用于治疗晚期GIST缩写的TKIs的关键试验
目前,我们仍然没有一种具有泛KIT抑制活性的批准治疗方法,可以克服携带KIT中已知发生的大量次级突变的亚克隆的肿瘤间和肿瘤内异质性。尽管有科学理由建议交替或联合使用激酶*制剂抑**,但过去的试验没有产生有希望的结果[29,30]。
除四线利普替尼外的治疗方法仍不清楚且具有挑战性。一项针对所有标准TKI均进展的转移性GIST患者的III期安慰剂对照试验(RIGHT 研究)表明,恢复伊马替尼可能会带来益处,因为肿瘤可能继续携带对激酶*制剂抑**敏感的克隆[31],这可能是一种合理的治疗方法,特别是如果患者无法参加其他临床试验,并提出了患者是否应该继续接受TKI的问题。
未来可能会结合循环肿瘤DNA(ctDNA)和肿瘤组织的下一代测序技术,不仅阐明突变异质性,而且阐明治疗中出现的新突变的进化,并可能具有治疗意义。此外,ctDNA检测可以识别使用组织分析未发现的KIT突变,这些突变可以解决肿瘤内异质性。一旦可以确定驱动因素的继发性突变,额外的靶向治疗可能会有所帮助[34]。

Amitesh C. Roy医生
未满足需求领域:野生型GIST
缺乏KIT或PDGFR突变的GIST通常被称为野生型GIST,通常对伊马替尼耐药。这些肿瘤亚组中遗传变化的例子包括SDH异常或RAS-RAF-MEK-ERK途径的激活。缺乏SDH的GIST是儿科人群GIST的主要原因。这组患者在编码SDH复合物的基因中具有种系致病性改变,导致SDH功能障碍,这反过来又容易导致GIST的发展[35,36]。
尽管野生型GIST具有独特的生物学特征,但通常与KIT或PDGFR突变的GIST包含在相同的临床试验中。这种方法使得定义治疗策略具有挑战性。鉴于这种持续未满足的需求,未来的国际合作需要为这部分患者确定更好的治疗策略。
小结
在首次伊马替尼研究发表二十年后,已经批准了几种治疗方案用于晚期GIST的治疗。GIST的分子检测对于为患者选择最佳治疗方案至关重要,未来研究的关键领域涉及克服多克隆耐药性和野生型GIST的管理。
参考文献
上下滑动查看更多内容
1. Demetri GD, von Mehren M, Blanke CD, et al. Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N Engl J Med. 2002;347(7):472-480.
2. Casali PG, Abecassis N, Aro HT, et al; ESMO Guidelines Committee and EURACAN. Gastrointestinal stromal tumours: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018;29(Suppl 4):iv68-iv78.
3. Dagher R, Cohen M, Williams G, et al. Approval summary: imatinib mesylate in the treatment of metastatic and/or unresectable malignant gastrointestinal stromal tumors. Clin Cancer Res. 2002;8(10):3034-3038.
4. U.S. Food and Drug Administration. FDA approves avapritinib for gastrointestinal stromal tumor with a rare mutation. Accessed November 16, 2022.
5. Verweij J, Casali PG, Zalcberg J, et al. Progression-free survival in gastrointestinal stromal tumours with high-dose imatinib: randomised trial. Lancet. 2004;364(9440):1127-1134.
6. Casali PG, Zalcberg J, Le Cesne A, et al; European Organisation for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group, Italian Sarcoma Group, and Australasian Gastrointestinal Trials Group. Ten-year progression-free and overall survival in patients with unresectable or metastatic GI stromal tumors: long-term analysis of the European Organisation for Research and Treatment of Cancer, Italian Sarcoma Group, and Australasian Gastrointestinal Trials Group intergroup phase III randomized trial on imatinib at two dose levels. J Clin Oncol. 2017;35(15):1713-1720.
7. Blanke CD, Rankin C, Demetri GD, et al. Phase III randomized, intergroup trial assessing imatinib mesylate at two dose levels in patients with unresectable or metastatic gastrointestinal stromal tumors expressing the kit receptor tyrosine kinase: S0033. J Clin Oncol. 2008;26(4):626-632.
8. Demetri GD, van Oosterom AT, Garrett CR, et al. Efficacy and safety of sunitinib in patients with advanced gastrointestinal stromal tumour after failure of imatinib: a randomised controlled trial. Lancet. 2006;368(9544):1329-1338.
9. George S, Blay JY, Casali PG, et al. Clinical evaluation of continuous daily dosing of sunitinib malate in patients with advanced gastrointestinal stromal tumour after imatinib failure. Eur J Cancer. 2009;45(11):1959-1968.
10. Demetri GD, Reichardt P, Kang YK, et al; GRID study investigators. Efficacy and safety of regorafenib for advanced gastrointestinal stromal tumours after failure of imatinib and sunitinib (GRID): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381(9863):295-302.
11. Blay JY, Serrano C, Heinrich MC, et al. Ripretinib in patients with advanced gastrointestinal stromal tumours (INVICTUS): a double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 2020;21(7):923-934.
12. Heinrich MC, Jones RL, von Mehren M, et al. Avapritinib in advanced PDGFRA D842V-mutant gastrointestinal stromal tumour (NAVIGATOR): a multicentre, open-label, phase 1 trial. Lancet Oncol. 2020;21(7):935-946.
13. Lasota J, Miettinen M. Clinical significance of oncogenic KIT and PDGFRA mutations in gastrointestinal stromal tumours. Histopathology. 2008;53(3):245-266.
14. Kelly CM, Gutierrez Sainz L, Chi P. The management of metastatic GIST: current standard and investigational therapeutics. J Hematol Oncol. 2021;14(1):2.
15. Gastrointestinal Stromal Tumor Meta-Analysis Group (MetaGIST). Comparison of two doses of imatinib for the treatment of unresectable or metastatic gastrointestinal stromal tumors: a meta-analysis of 1,640 patients. J Clin Oncol. 2010;28(7):1247-1253.
16. Corless CL, Barnett CM, Heinrich MC. Gastrointestinal stromal tumours: origin and molecular oncology. Nat Rev Cancer. 2011;11(12):865-878.
17. Corless CL, Schroeder A, Griffith D, et al. PDGFRA mutations in gastrointestinal stromal tumors: frequency, spectrum and in vitro sensitivity to imatinib. J Clin Oncol. 2005;23(23):5357-5364.
18. Heinrich MC, Corless CL, Duensing A, McGreevey L, Chen CJ, Joseph N, et al. PDGFRA activating mutations in gastrointestinal stromal tumors. Science. 2003;299(5607):708-710.
19. Indio V, Astolfi A, Tarantino G, et al. Integrated molecular characterization of gastrointestinal stromal tumors (GIST) harboring the rare D842V mutation in PDGFRA gene. Int J Mol Sci. 2018;19(3):732.
20. Kang YK, George S, Jones RL, Rutkowski P, Shen L, Mir O, et al. Avapritinib versus regorafenib in locally advanced unresectable or metastatic GI stromal tumor: a randomized, open-label phase III study. J Clin Oncol. 2021;39(28):3128-3139.
21. Schaefer IM, DeMatteo RP, Serrano C. The GIST of advances in treatment of advanced gastrointestinal stromal tumor. Am Soc Clin Oncol Educ Book. 2022;42:1-15.
22. Blanke CD, Demetri GD, von Mehren M, et al. Long-term results from a randomized phase II trial of standard- versus higher-dose imatinib mesylate for patients with unresectable or metastatic gastrointestinal stromal tumors expressing KIT. J Clin Oncol. 2008;26(4):620-625.
23. Barnett CM, Heinrich MC. Management of tyrosine kinase inhibitor-resistant gastrointestinal stromal tumors. Am Soc Clin Oncol Educ Book. 2012;32:663-668.
24. Serrano C, Mariño-Enríquez A, Tao DL, et al. Complementary activity of tyrosine kinase inhibitors against secondary kit mutations in imatinib-resistant gastrointestinal stromal tumours. Br J Cancer. 2019;120(6):612-620.
25. QINLOCK® (ripretinib) clinical overview. Published online 2021.
26. U.S. Food and Drug Administration. FDA approves ripretinib for advanced gastrointestinal stromal tumor. Accessed November 16, 2022.
27. Nemunaitis J, Bauer S, Blay JY, et al. Intrigue: phase III study of ripretinib versus sunitinib in advanced gastrointestinal stromal tumor after imatinib. Future Oncol. 2020;16(1):4251-4264.
28. Bauer S, Jones RL, Blay JY, Gelderblom H, George S, Schöffski P, et al. Ripretinib versus sunitinib in patients with advanced gastrointestinal stromal tumor after treatment with imatinib (INTRIGUE): a randomized, open-label, phase III trial. J Clin Oncol. Published online August 10, 2022.
29. Serrano C, Fletcher JA. Overcoming heterogenity in imatinib-resistant gastrointestinal stromal tumor. Oncotarget. 2019;10(59):6286-6287.
30. Yip D, Zalcberg JR, Blay JY, et al. ALT-GIST: randomized phase II trial of imatinib alternating with regorafenib versus imatinib alone for the first-line treatment of metastatic gastrointestinal stromal tumor (GIST). J Clin Oncol. 2019;37(suppl 15; abstr 11023):11023.
31. Kang YK, Ryu MH, Yoo C, et al. Resumption of imatinib to control metastatic or unresectable gastrointestinal stromal tumours after failure of imatinib and sunitinib (RIGHT): a randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 2013;14(12):1175-1182.
32. Xu H, Chen L, Shao Y, et al. Clinical application of circulating tumor DNA in the genetic analysis of patients with advanced GIST. Mol Cancer Ther. 2018;17(1):290-296.
33. Arshad J, Roberts A, Ahmed J, et al. Utility of circulating tumor DNA in the management of patients with GI stromal tumor: analysis of 243 patients. JCO Precis Oncol. 2020;4:66-73.
34. Schwartz GK. Ripretinib and the failure to advance GI stromal tumor therapy in the age of precision medicine. J Clin Oncol. Published online September 2, 2022.
35. Pasini B, McWhinney SR, Bei T, et al. Clinical and molecular genetics of patients with the Carney-Stratakis syndrome and germline mutations of the genes coding for the succinate dehydrogenase subunits SDHB, SDHC, and SDHD. Eur J Hum Genet. 2008;16(1):79-88.
36. Gaal J, Stratakis CA, Carney JA, et al. SDHB immunohistochemistry: a useful tool in the diagnosis of Carney-Stratakis and Carney triad gastrointestinal stromal tumors. Mod Pathol. 2011;24(1):147-151.